Option (a) is the correct answer.
Explanation:
Atomic number of argon is 18 and its electronic configuration is ![[Ne] 3s^{2}3p^{6}](/tpl/images/0264/8873/f7d86.png) . Hence, there are eight electrons present in the outermost shell of argon.
. Hence, there are eight electrons present in the outermost shell of argon.
Atomic number of beryllium is 4 and its electronic configuration is 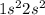 . Only 2 electrons are present in its outermost shell.
. Only 2 electrons are present in its outermost shell.
Atomic number of chromium is 24 with electronic configuration ![[Ar] 4s^{1} 3d^{5}](/tpl/images/0264/8873/e2e7b.png) . There are only 5 valence electrons present in it.
. There are only 5 valence electrons present in it.
Atomic number of oxygen is 8 and its electronic configuration is 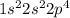 . Hence, there are only 6 electrons present in its valence shell.
. Hence, there are only 6 electrons present in its valence shell.
Thus, we can conclude that out of the given options argon element has eight valence electrons.

























