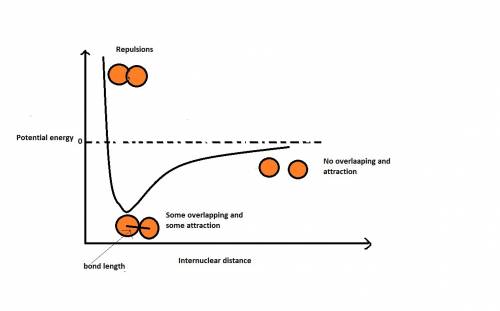
Bond length is the distance between the centers of two bonded atoms. on the potential energy curve, the bond length is the internuclear distance between the two atoms when the potential energy of the system reaches its lowest value. given that the atomic radii of h and cl are 25.0 cm and 100. pm , respectively, predict the bond length of the hcl molecule.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Write the balanced equation for a reaction between aqueous nitric acid (hno3) and solid lithium metal (this is a single replacement reaction)
Answers: 1

Chemistry, 22.06.2019 07:20
After watching the video "zinc strip in copper nitrate solution", and reading the instructions, click on the link labeled "start" just below the drawing of the pencil tip. follow the direction to complete the 3x3 grid. answer the below questions for the portion of the activity in which sn(s) is placed in agno3(aq)
Answers: 1

Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 09:30
Right anwser gets marked brainliest newton's discovery concerning how fast an object will change speed is the: 1st law 2nd law 3rd law universal gravitation
Answers: 1
You know the right answer?
Bond length is the distance between the centers of two bonded atoms. on the potential energy curve,...
Questions



Mathematics, 22.04.2020 16:46

Biology, 22.04.2020 16:47

Biology, 22.04.2020 16:47




English, 22.04.2020 16:47






History, 22.04.2020 16:47

Computers and Technology, 22.04.2020 16:47

English, 22.04.2020 16:47

Computers and Technology, 22.04.2020 16:47