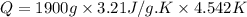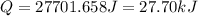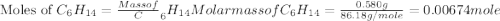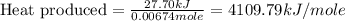Chemistry, 19.11.2019 04:31 billgray2571
Asample of hexane (c6h14) has a mass of 0.580 g. the sample is burned in a bomb calorimeter that has a mass of 1.900 kg and a specific heat of 3.21 j/gik. what amount of heat is produced during the combustion of hexane if the temperature of the calorimeter increases by 4.542 k?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Arock can be broken down into different kinds of substances by physical processes. no chemical reactions are needed to separate different parts of a rock into pure substances. this is because a rock is a(n)
Answers: 1

Chemistry, 22.06.2019 00:00
Which type of bonding involves the complete transfer of a valence electron from a less electrogrative atom to a more electronegative one
Answers: 1

Chemistry, 22.06.2019 13:30
The atomic number, or number, is the described as the number of in the nucleus of an chemical element.
Answers: 1

Chemistry, 22.06.2019 18:30
Asample of hydrated tin (ii) chloride (sncl2) has a mass of 4.90 g. when it is dehydrated, it has a mass of 4.10 g. which is the correct chemical formula for the hydrate? sncl2•2h2o sncl2•4h2o sncl2•6h2o
Answers: 2
You know the right answer?
Asample of hexane (c6h14) has a mass of 0.580 g. the sample is burned in a bomb calorimeter that has...
Questions


Social Studies, 20.10.2021 06:20

History, 20.10.2021 06:20

Mathematics, 20.10.2021 06:20

Mathematics, 20.10.2021 06:20

Computers and Technology, 20.10.2021 06:20

Mathematics, 20.10.2021 06:20



Mathematics, 20.10.2021 06:20





Mathematics, 20.10.2021 06:20

History, 20.10.2021 06:20


Social Studies, 20.10.2021 06:20

Mathematics, 20.10.2021 06:20

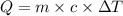
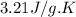
 = change in temperature = 4.542 K
= change in temperature = 4.542 K