Which is the formula mass of na2so4?
119.06 amu
125.98 amu
142.05 amu
174.1...

Chemistry, 04.01.2020 20:31 ThousandSeas9381
Which is the formula mass of na2so4?
119.06 amu
125.98 amu
142.05 amu
174.12 amu

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:30
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1

Chemistry, 22.06.2019 06:40
Which statement is usually true about the relationship between activation energy and reaction rates? low activation energy barriers result in low rates. high activation energy barriers result in low rates. low activation energy barriers result in no reaction. high activation energy barriers result in no reaction.
Answers: 3

Chemistry, 22.06.2019 08:00
Define dew point. i am writing this part to be able to ask the question
Answers: 1

Chemistry, 22.06.2019 08:30
Identify one disadvantage to each of the following models of electron configuration: -dot structures -arrow and line diagrams -written electron configurations type in your answer below. (answer) -dot structures do not show the distribution of electrons in orbitals and take up a lot of space. -arrow and line diagrams take up a lot of space and make it difficult to count electrons. -written configurations make it easy to lose count of electrons and do not show the distribution of electrons in orbitals.
Answers: 3
You know the right answer?
Questions



Biology, 01.10.2019 19:30

English, 01.10.2019 19:30



History, 01.10.2019 19:30





Health, 01.10.2019 19:30

Mathematics, 01.10.2019 19:30

Computers and Technology, 01.10.2019 19:30


Mathematics, 01.10.2019 19:30

English, 01.10.2019 19:30

Physics, 01.10.2019 19:30


English, 01.10.2019 19:30

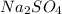 = 2(22.99)+32.065+4(15.999)= 142.05amu
= 2(22.99)+32.065+4(15.999)= 142.05amu

