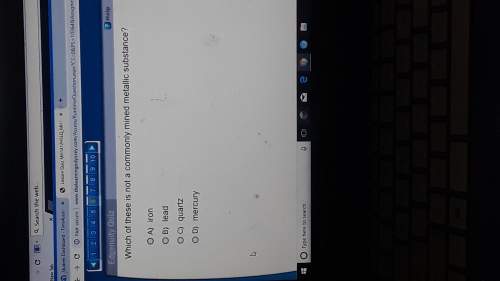
The ionization energy of an element is
a. a measure of its mass
b. the energy required...

Chemistry, 18.08.2019 18:30 queenpaige2015
The ionization energy of an element is
a. a measure of its mass
b. the energy required to remove an electron from the element in its gaseous state
c. the energy released by the element in forming an ionic bond
d. the energy released by the element upon receiving an additional electron
e. none of the above

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 16:30
Correct relationship between molecular formula and empirical formula
Answers: 1


Chemistry, 22.06.2019 18:20
Which reason best explains why metals are malleable? a)because they have delocalized electrons b)because they have localized electrons c)because they have ionic bonds d)because they have rigid bonds
Answers: 2

Chemistry, 22.06.2019 21:30
How many oxygen atoms are there in 3.15 moles of hcl manganese (iv) oxide, mno2
Answers: 2
You know the right answer?
Questions


Spanish, 10.03.2021 20:30



Mathematics, 10.03.2021 20:30




Geography, 10.03.2021 20:30

Mathematics, 10.03.2021 20:30



Computers and Technology, 10.03.2021 20:30







Mathematics, 10.03.2021 20:30