What is oxidized and what is reduced in this reaction?
n2h4(l) + 2h2o2(l) → n2(g) + 4h2...


Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:30
What is the relation between concentration of reactants and the rate of chemical reaction?
Answers: 1

Chemistry, 22.06.2019 04:00
Which of the following ocean acidification? are the most likely side effects of a ph less than 7.0 in the ocean b. more metal salts altering the ocean chemistry c. dissolution of the shells of marine organisms d. both a & b e. all of the above.
Answers: 3

Chemistry, 22.06.2019 12:10
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3

Chemistry, 22.06.2019 19:50
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1
You know the right answer?
Questions

Mathematics, 12.12.2019 21:31

Mathematics, 12.12.2019 21:31

History, 12.12.2019 21:31

Mathematics, 12.12.2019 21:31





Mathematics, 12.12.2019 22:31





Mathematics, 12.12.2019 22:31

Mathematics, 12.12.2019 22:31

Mathematics, 12.12.2019 22:31


History, 12.12.2019 22:31


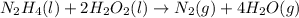
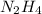 gives
gives 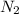 gas which means nitrogen is getting oxidized.Whereas removal of oxygen from
gas which means nitrogen is getting oxidized.Whereas removal of oxygen from 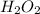 gives
gives 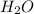 gas which means that oxygen is getting reduced.
gas which means that oxygen is getting reduced.


