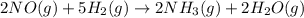Chemistry, 07.12.2019 16:31 andygomez1312M
The balanced chemical equation, 2no (g) + 5h2 (g) (arrow pointing this way > > ) 2nh3 (g) + 2h2o (g), can be expressed in words as:
nitrogen dioxide gas plus hydrogen gas yields ammonia gas plus water
nitrogen monoxide gas plus hydrogen gas yields ammonia gas plus water vapor
nickel monoxide gas plus hydrogen gas yields nitrogen trihydride plus water
nitrogen oxide gas plus hydrogen gas yields ammonia gas plus water vapor
i think it's either b, or d. i can't figure out which one it is though. .

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:40
During trial 2, what allowed you to determine that aluminum was the limiting reactant? check all that apply. all of the copper dissolved. all of the aluminum dissolved. the solution turned clear. the number of grams of copper(ii) chloride used in the reaction was greater than the number of grams of aluminum. the molar ratio of copper(ii) chloride to aluminum was greater than 3: 2, the equation’s molar ratio.
Answers: 2


Chemistry, 23.06.2019 01:50
Ablock of aluminum is dropped into a graduated cylinder with an initial volume of water at 75ml and the volumes rises to 90ml. if the block has a mass of 40.5 g what is its density ?
Answers: 1

Chemistry, 23.06.2019 03:30
In general metals get as you move from left to right across the periodic table.
Answers: 1
You know the right answer?
The balanced chemical equation, 2no (g) + 5h2 (g) (arrow pointing this way > > ) 2nh3 (g) + 2h...
Questions





Mathematics, 24.04.2020 03:28


Biology, 24.04.2020 03:28



Biology, 24.04.2020 03:28

Mathematics, 24.04.2020 03:28

History, 24.04.2020 03:28

Mathematics, 24.04.2020 03:28

Advanced Placement (AP), 24.04.2020 03:28



Advanced Placement (AP), 24.04.2020 03:28