

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:30
Match the following vocabulary terms to their definitions. 1. amount of energy required to change 1 gram of material from the solid to the liquid state at its melting point 2. a measure of the kinetic energy of the particles of a substance 3. the amount of heat energy required to raise the temperature of 1 gram of liquid water from 14.5°c to 15.5°c 4. amount of energy required to change 1 gram of material from the liquid to the gaseous state at its boiling point 5. the amount of energy required to change 1 gram of a substance 1°c a. temperature b. latent heat of vaporization c. latent heat of fusion d. calorie e. specific heat
Answers: 1

Chemistry, 22.06.2019 06:30
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1

Chemistry, 22.06.2019 22:00
How many moles of no2 will form when 3.3 moles of cu are reacted with excess hno3?
Answers: 3

Chemistry, 23.06.2019 04:40
6) (a) calculate the absorbance of the solution if its concentration is 0.0278 m and its molar extinction coefficient is 35.9 l/(mol cm). the depth of the cell is 5 mm. (b) what is the %t? (7) calculate the absorbance of the solution if the transmitted light intensity is 70% of the initial light beam intensity
Answers: 1
You know the right answer?
How many moles of oxygen are needed to completely react with 9.5 grams of sodium...
Questions





Mathematics, 09.11.2019 02:31


Social Studies, 09.11.2019 02:31


English, 09.11.2019 02:31

Mathematics, 09.11.2019 02:31

Biology, 09.11.2019 02:31





Physics, 09.11.2019 02:31

Mathematics, 09.11.2019 02:31


World Languages, 09.11.2019 02:31

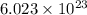 of particles.
of particles.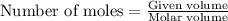
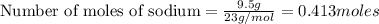
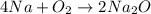
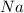 combine completely with 1 mole of
combine completely with 1 mole of 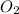 to give 2 moles of
to give 2 moles of 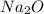
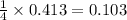 moles of
moles of 

