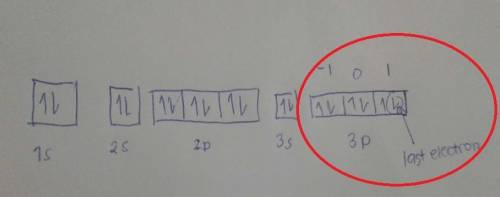Answers: 1
Another question on Chemistry

Chemistry, 23.06.2019 06:40
1.) which of the following is a molecule but not a compound? a.he b.f2 c.h2o d.ch4 2.) what is a physical combination of substances? a.a compound b.a molecule c.a mixture d.an element 3.) what is a chemical combination of substances? a.a compound b.an atom c.a mixture d.an element 4.) what is the relationship between the solute and solvent in a solution? a.they form a compound b.they form a mixture c.they form molecules d.they form chemical bonds 5.) the gases in air dissolve in water. what would be one way to reduce the amount of a gas dissolved in water? a.add more water b.reduce the air pressure c.increase the air pressure d.stir the water 6.) how would you determine the solubility of a substance? a.find how well it dissolved various substances. b.find the mass and the volume of the substance. c.find the temperature at which the substance evaporated. d.find how much i was able to dissolve in a solute. 7.) the periodic table organizes all of the kinds of a.molecules. b.compounds. c.atoms. d.ions. 8.)what distinguishes two substances combined to become a compound vs. two substances combined to become a mixture? a.whether they can be easily separated b.whether they chemically bond together c.whether they both are visible d.whether they are heterogeneous 9.) the principle components of air are: n2 78% o2 21% ar 0.95% co2 0.038% this is a solution of a.molecules and atoms. b.molecules. c.compounds and molecules. d.atoms.
Answers: 1

Chemistry, 23.06.2019 10:30
Silver is a white metal that is an excellent conductor. silver tarnishes when exposed to air and light. the density of silver is 10.49 g/cm3. the melting point is 962oc and the boiling point is 2000oc. a chemical property of silver is
Answers: 3

Chemistry, 23.06.2019 11:40
An electron moved from a lower energy level to a higher energy level. what most likely happened during the transition? a random amount of light was released. a fixed amount of energy was absorbed. a fixed amount of energy was released. a random amount of light was absorbed.
Answers: 1

Chemistry, 23.06.2019 15:30
Ms. sullivan's class is designing a miniature roller coaster. after first setting it up, they find that it didn't have enough speed to go through the loop de loop. instead of taking out the loop, they decide to increase the height of the first hill the coaster goes down, and this adds the speed needed to make it through the loop. what would be a drawback to this plan in real life? a) no one likes loop de loops. b) there is no downside to this plan. c) a higher hill means a scarier ride. d) a higher hill takes more time and material to build.
Answers: 1
You know the right answer?
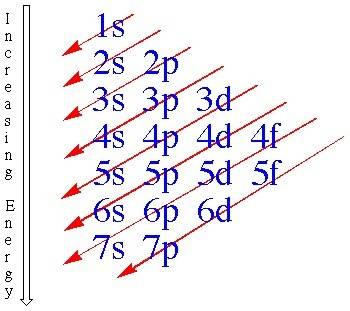
Give the set of four quantum numbers that could represent the electron lost to form the k ion from t...
Questions

Biology, 20.08.2019 13:10

Geography, 20.08.2019 13:10


Mathematics, 20.08.2019 13:10


Mathematics, 20.08.2019 13:10

Physics, 20.08.2019 13:10


Mathematics, 20.08.2019 13:10

Mathematics, 20.08.2019 13:10

Mathematics, 20.08.2019 13:10

English, 20.08.2019 13:10



History, 20.08.2019 13:10





Mathematics, 20.08.2019 13:10