
Chemistry, 01.09.2019 02:30 nulledcracker12
Consider the balanced chemical equation that follows. you are asked to determine how many moles of water you can produce from 4.0 mol of hydrogen and excess oxygen. (excess oxygen means that so much oxygen is available it will not run out.) which of the numbers that appear in the balanced chemical equation below are used to perform this calculation? 2h2(g)+o2(g)→2h2o(l)

Answers: 1
Another question on Chemistry




Chemistry, 22.06.2019 11:30
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2
You know the right answer?
Consider the balanced chemical equation that follows. you are asked to determine how many moles of w...
Questions





History, 16.10.2019 00:40





Social Studies, 16.10.2019 00:40

History, 16.10.2019 00:40


History, 16.10.2019 00:40


Social Studies, 16.10.2019 00:40


Health, 16.10.2019 00:40


Health, 16.10.2019 00:40

History, 16.10.2019 00:40


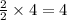 moles of water
moles of water

