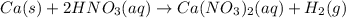Chemistry, 31.08.2019 15:30 birdman37361
Ca(s)+2hno3(aq)→ca(no3)2(aq)+h2(g) identify the oxidizing agent.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:30
Write the chemical formula for a compound that is made of an element from group 1 and an element from group 17
Answers: 1

Chemistry, 23.06.2019 04:20
The equation below shows a chemical reaction. a + b + heat —> c + d according to the law of conservation of energy, which statement is true? a. the reactants absorb heat because they have less energy than the products. b. the products release heat because they have more energy than the reactants. c. the reactants generate heat because they have more energy than the products. d. the products require heat to form because they have less energy than the reactants.
Answers: 1

Chemistry, 23.06.2019 04:40
[01.07]what is the answer to the problem: 101 g + 25.01 g + 5.05 g? 131.06 g 131.1 g 131 g 130 g
Answers: 1

Chemistry, 23.06.2019 08:00
Determine the number of moles of air present in 1.35 l at 750 torr and 17.0°c. which equation should you use? n=pv/rt what is the number of moles present? ⇒ 0.056 mol a sample of n2 gas occupying 800.0 ml at 20.0°c is chilled on ice to 0.00°c. if the pressure also drops from 1.50 atm to 1.20 atm, what is the final volume of the gas? which equation should you use? v2= p1v1t2/p2t1 what is the final volume of the gas? ⇒ 932 ml these are the answers
Answers: 1
You know the right answer?
Ca(s)+2hno3(aq)→ca(no3)2(aq)+h2(g) identify the oxidizing agent....
Questions

Mathematics, 20.09.2020 18:01


Mathematics, 20.09.2020 18:01

Mathematics, 20.09.2020 18:01

English, 20.09.2020 18:01








Mathematics, 20.09.2020 18:01

Social Studies, 20.09.2020 18:01

Biology, 20.09.2020 18:01

Chemistry, 20.09.2020 18:01



Mathematics, 20.09.2020 18:01