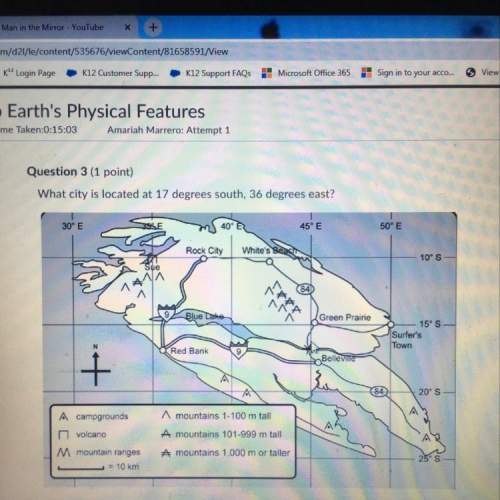
What is the correct equilibrium expression for the dissociation of the base pyridine:
C5H5N + H2O ⇋ C5H5NH+ + OH-
A. Kb = [C5H5NH+][OH-] / [C5H5N]
B. Kb = [C5H5N][OH-] / [C5H5NH+][H2O]
C .Kb = [C5H5NH+][OH-] / [C5H5N][H2O]
D .Kb = [C5H5NH+][C5H5N] / [OH-]
E. Kb = [C5H5N][OH-] / [C5H5NH+]

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:00
What is important to study for nios grade 12 chemistry? i have only one month left.
Answers: 2

Chemistry, 22.06.2019 04:00
Write the empirical chemical formula of calcium with a mass percent of 38.8, phosphorus with a mass percent of 20.0, and oxygen with a mass percent of 41.3.
Answers: 1

Chemistry, 22.06.2019 17:00
Which property of a rock remains unchanged by mechanical weathering? a. total surface area b. size and shape c. mineral composition d. sharpness
Answers: 1

Chemistry, 22.06.2019 20:30
Which of the following is not true about the atomic model of substances?
Answers: 1
You know the right answer?
What is the correct equilibrium expression for the dissociation of the base pyridine:
C5H5N + H2O...
Questions


Mathematics, 20.07.2019 14:00





Mathematics, 20.07.2019 14:00




History, 20.07.2019 14:00

Mathematics, 20.07.2019 14:00

Computers and Technology, 20.07.2019 14:00


History, 20.07.2019 14:00

Social Studies, 20.07.2019 14:00


Mathematics, 20.07.2019 14:00

Mathematics, 20.07.2019 14:00

Biology, 20.07.2019 14:00