2 H202 - 2 H2O + O2

Chemistry, 22.04.2021 04:40 cjasmine626
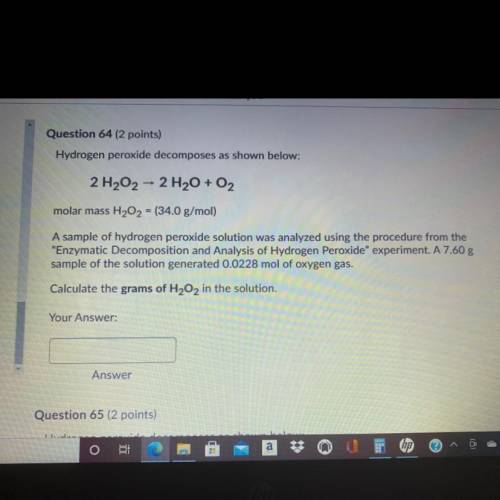
Question 64 (2 points)
Hydrogen peroxide decomposes as shown below:
2 H202 - 2 H2O + O2
molar mass H202 = (34.0 g/mol)
A sample of hydrogen peroxide solution was analyzed using the procedure from the
"Enzymatic Decomposition and Analysis of Hydrogen peroxide" experiment. A 7.60 g
sample of the solution generated 0.0228 mol of oxygen gas.
Calculate the grams of H2O2 in the solution.
Your
Answer

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:30
**40** points asapessay questions (10 points possible) clear image of next, create your own scenario. it can be one of your own real experiences or one you make up. use imagery in your writing to give your instructor a the setting and an action taking pace in your writing explain the structure and functions of the skin at work in your scenario. !
Answers: 3

Chemistry, 22.06.2019 09:00
Which process does not require the presence of a physical substance in order to transfer heat? air in the atmosphere is heated by the ground. this warm air then rises, and cooler air falls. this is an example of what type of process? how is conduction different from radiation?
Answers: 1

Chemistry, 22.06.2019 10:30
Skills of homo sapiens were found an excavation. the skulls were preserved because the bodies were frozen. so, these fossils are (blank) fossils.the image shows the evolution of skulls beginning 2 to 3 million years ago. based on the image, modern human skulls(blank) ape skulls.
Answers: 1

Chemistry, 22.06.2019 11:00
An object becomes electrically charged when: electrons are created in it electrons from it are destroyed electrons are transferred to it protons from it are destroyed protons are created in it
Answers: 1
You know the right answer?
Question 64 (2 points)
Hydrogen peroxide decomposes as shown below:
2 H202 - 2 H2O + O2
2 H202 - 2 H2O + O2
Questions


Spanish, 11.07.2019 20:40


English, 11.07.2019 20:40


Social Studies, 11.07.2019 20:40


Mathematics, 11.07.2019 20:40

Mathematics, 11.07.2019 20:40

Chemistry, 11.07.2019 20:40

Mathematics, 11.07.2019 20:40

English, 11.07.2019 20:40


Social Studies, 11.07.2019 20:40

History, 11.07.2019 20:40

Mathematics, 11.07.2019 20:40



Biology, 11.07.2019 20:40



