
Chemistry, 22.04.2021 20:40 CHEVYWADDELL
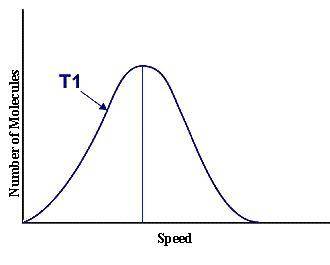
This graph represents a population of molecules in a gas versus the distribution of the average velocity(speed) of its molecules in that population. Assume all molecules to be of the same mass. In reading the graph, it is important to note three things. One, is the most probable speed is at the peak of the curve. Secondly, the most probable speed increases as the temperature increases (so shift to the right), and the distribution broadens as it increases.
On the graph, indicate the average kinetic energy of the population.
Explain your answer.
What part of the graph indicates the temperature of the sample?
Explain your answer.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1


Chemistry, 22.06.2019 17:40
How much heat is added if 0.814g of water increase in temperature by 0.351 degree c?
Answers: 3
You know the right answer?
This graph represents a population of molecules in a gas versus the distribution of the average velo...
Questions

Spanish, 29.05.2020 00:08

English, 29.05.2020 00:08

Mathematics, 29.05.2020 00:08


Chemistry, 29.05.2020 00:08


History, 29.05.2020 00:08



Mathematics, 29.05.2020 00:08



Mathematics, 29.05.2020 00:57

Mathematics, 29.05.2020 00:57

Mathematics, 29.05.2020 00:57

Mathematics, 29.05.2020 00:57

Mathematics, 29.05.2020 00:57

Mathematics, 29.05.2020 00:57


Mathematics, 29.05.2020 00:57



