PLEASEEE HELP A chemist mixed two substances
together: a blue powder with no smell
and a col...

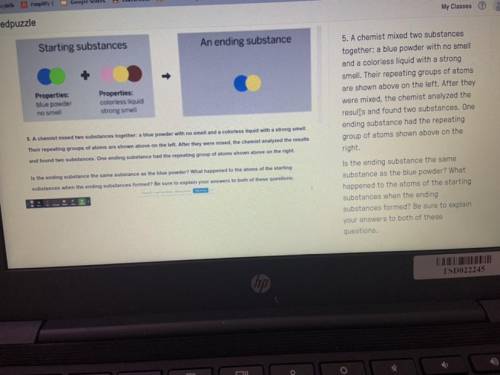
PLEASEEE HELP A chemist mixed two substances
together: a blue powder with no smell
and a colorless liquid with a strong
smell. Their repeating groups of atoms
are shown above on the left. After they
were mixed, the chemist analyzed the
results and found two substances. One
ending substance had the repeating
group of atoms shown above on the
right.
Is the ending substance the same
substance as the blue powder? What
happened to the atoms of the starting
substances when the ending
substances formed? Be sure to explain
your answers to both of these

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:00
During chemistry class, carl performed several lab tests on two white solids. the results of three tests are seen in the data table. based on this data, carl has concluded that substance b must have bonds.
Answers: 2

Chemistry, 22.06.2019 05:30
What is the mass defect of a mole of nuclei with 1.8 x 10^15 j/mol binding energy?
Answers: 1

Chemistry, 22.06.2019 13:40
Can someone me with 6 to 10 plz this is for masteries test.
Answers: 1

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3
You know the right answer?
Questions




Mathematics, 30.03.2020 06:00

English, 30.03.2020 06:00

Mathematics, 30.03.2020 06:00

Biology, 30.03.2020 06:00

Health, 30.03.2020 06:00

Mathematics, 30.03.2020 06:00

Mathematics, 30.03.2020 06:00

Mathematics, 30.03.2020 06:00

Mathematics, 30.03.2020 06:00

Physics, 30.03.2020 06:00

Arts, 30.03.2020 06:00

Mathematics, 30.03.2020 06:00



Mathematics, 30.03.2020 06:00

Mathematics, 30.03.2020 06:00



