
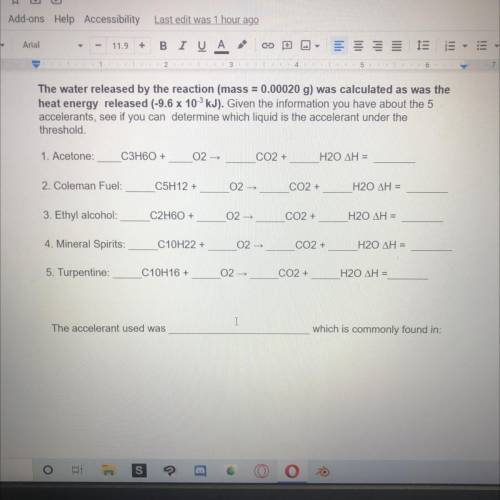
The water released by the reaction (mass = 0.00020 g) was calculated as was the
heat energy released (-9.6 x 10 kJ). Given the information you have about the 5
accelerants, see if you can determine which liquid is the accelerant under the
threshold.
1. Acetone:
C3H60+
02-
CO2 +
H20 ΔΗ =
2. Coleman Fuel:
C5H12 +
02-
CO2 +
H20 ΔΗ =
3. Ethyl alcohol
C2H60 +
02 -
CO2 +
H20 ΔΗ =
4. Mineral Spirits:
C10H22 +
02-
CO2 +
H20 AH =
5. Turpentine:
C10H16 +
02 -
CO2 +
H20 ΔΗ =
The accelerant used was
I
which is commonly found in:

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:00
If the particles in a sample of matter have an orderly arrangement and move only in place, the sample is a
Answers: 1

Chemistry, 21.06.2019 23:00
Write a brief passage describing a neutral atom of nitrogen-14 (n-14). describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. use the periodic table to you. 14 protons and eletrons since its a neutral atom
Answers: 1

Chemistry, 22.06.2019 10:20
In a reaction equation, where are the products located? a.) above the arrow b.) to the right of the arrow c.) to the left of the arrow d.) below the arrow
Answers: 2

Chemistry, 22.06.2019 13:50
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1
You know the right answer?
The water released by the reaction (mass = 0.00020 g) was calculated as was the
heat energy releas...
Questions


Biology, 09.12.2019 06:31


Mathematics, 09.12.2019 06:31

Mathematics, 09.12.2019 06:31

Mathematics, 09.12.2019 06:31

English, 09.12.2019 06:31


History, 09.12.2019 06:31


Biology, 09.12.2019 06:31


Mathematics, 09.12.2019 06:31

Mathematics, 09.12.2019 06:31


Computers and Technology, 09.12.2019 06:31



Mathematics, 09.12.2019 06:31



