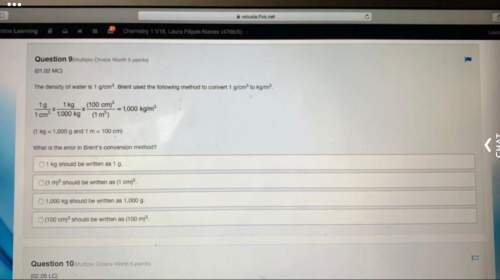
How many grams of hydrogen gas are produced if 6 moles of hydrochloric acid reacts with 2 mole of Al?
Extra Content
6HCl(aq) + 2Al(s) → 2AlCl3(s) + 3H2(g)
six moles of hydrochloric acid reacts with two moles of aluminum to produce 2 moles of aluminum chloride plus 3 moles of hydrogen gas.
A.
4.65 grams
B.
3 grams
C.
66.06 grams
D.
6.06 grams

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:00
In any energy conversion, some of the energy is lost to the environment as question 5 options: electrical energy potential energy sound energy thermal energy
Answers: 1

Chemistry, 22.06.2019 12:00
Which of the following is an example of physical change not a chemical change? a) a log gives off heat and light as it burns. b) a tree stores energy from the sun in its fruit. c) a penny lost in the grass slowly changes color. d) a water pipe freezes and cracks on a cold night.
Answers: 2

Chemistry, 22.06.2019 15:00
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1

You know the right answer?
How many grams of hydrogen gas are produced if 6 moles of hydrochloric acid reacts with 2 mole of Al...
Questions


Mathematics, 18.11.2020 01:00

Business, 18.11.2020 01:00



Mathematics, 18.11.2020 01:00


Social Studies, 18.11.2020 01:00

Chemistry, 18.11.2020 01:00




History, 18.11.2020 01:00

English, 18.11.2020 01:00


Mathematics, 18.11.2020 01:00

Mathematics, 18.11.2020 01:00

Social Studies, 18.11.2020 01:00

Mathematics, 18.11.2020 01:00

Mathematics, 18.11.2020 01:00