
Chemistry, 29.04.2021 06:30 Arielledt10
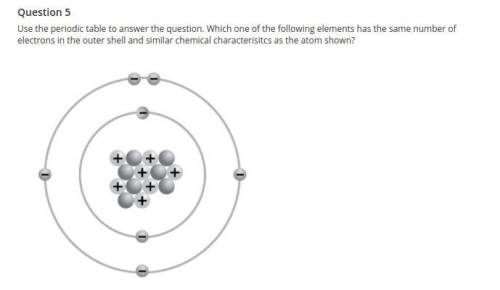
Use the periodic table to answer the question. Which one of the following elements has the same number of electrons in the outer shell and similar chemical characterisitcs as the atom shown?
a. I
b. K
c. S
d. Bi

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:00
What are the first two quantum numbers for the electrons located in subshell 4d? what are the first three quantum numbers for the electrons located in subshell 2s? how many electrons can be held in a sublevel l = 3? how many electrons can be held in the energy level n = 4? how many electrons in an atom can share the quantum numbers n = 4 and l = 3?
Answers: 1

Chemistry, 22.06.2019 12:30
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2

Chemistry, 22.06.2019 17:00
In a heat engine of 1000 j of heat enters the system and the piston does 500 j of work what is the final internal energy of the system if the inital energy was 2000 j we have to do all of these down here 1)write the equation 2)list out your know variables 3)plug the numbers into the equations 4)solve 5)write your solution statemtn that includes inital energuy and final energuy added
Answers: 1

Chemistry, 23.06.2019 01:00
Atoms contain subatomic particles called protons and neutrons. when these protons and neutrons spilt, a lot of energy is released
Answers: 3
You know the right answer?
Use the periodic table to answer the question. Which one of the following elements has the same numb...
Questions




World Languages, 22.06.2019 11:30

Mathematics, 22.06.2019 11:30



Mathematics, 22.06.2019 11:30



Mathematics, 22.06.2019 11:30


Mathematics, 22.06.2019 11:30


Mathematics, 22.06.2019 11:30


History, 22.06.2019 11:30


Mathematics, 22.06.2019 11:30



