What is the net What is the net cell potential for this electrochemical cell?cell
...

Chemistry, 29.04.2021 07:40 miner12924owhu4d
What is the net What is the net cell potential for this electrochemical cell?cell
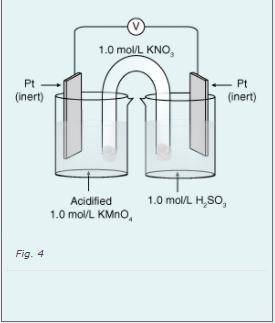

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:30
This element exists in adundance in the sun.explain how you would go about capturing sunlight.would this captured sunlight contain any of the element?
Answers: 1

Chemistry, 22.06.2019 10:30
Which of these is not an example of chemical weathering? a. iron-rich mineral rusting b. feldspar turning into clay c. limestone reacting with acid d. granite breaking up into sand
Answers: 1

Chemistry, 22.06.2019 13:00
These questions are based on the attached photo. the experiment is about burning magnesium metal with oxygen. 1. write the balanced chemical equation for the reaction you are performing. 2. calculate the mass of magnesium metal used in each trial. o trial 1: o trial 2: 3. calculate the actual yield of magnesium oxide for each trial. o trial 1: o trial 2: 4. magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. o trial 1: o trial 2: 5. determine the percent yield of mgo for your experiment for each trial. o trial 1: o trial 2: 6. determine the average percent yield of mgo for the two trials. your company currently uses a process with a similar cost of materials that has an average percent yield of 91 percent. if the average percent yield of this process is higher than that, this could save the company money. what is your recommendation to the company? support your recommendation using your data, calculations, and understanding of stoichiometry gathered from this lab.
Answers: 1

Chemistry, 22.06.2019 14:30
100 grams of molten lead (600°c) is used to make musket balls. if the lead shot is allowed to cool to room temperature (21°c), what is the change in entropy (in j/k) of the lead? (for the specific heat of molten and solid lead use 1.29 j/g⋅°c; the latent heat of fusion and the melting point of lead are 2.45 × 104 j/kg and 327°c, respectively.)
Answers: 1
You know the right answer?
Questions

Mathematics, 23.01.2020 23:31

English, 23.01.2020 23:31

History, 23.01.2020 23:31


History, 23.01.2020 23:31


Health, 23.01.2020 23:31

Mathematics, 23.01.2020 23:31

Social Studies, 23.01.2020 23:31

History, 23.01.2020 23:31

History, 23.01.2020 23:31

Mathematics, 23.01.2020 23:31


Biology, 23.01.2020 23:31

Mathematics, 23.01.2020 23:31

Mathematics, 23.01.2020 23:31


Mathematics, 23.01.2020 23:31

Business, 23.01.2020 23:31

Chemistry, 23.01.2020 23:31



