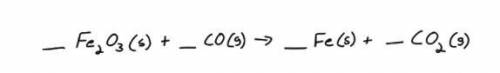Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 13:00
Which of the following are good traits of a hypothesis? it will be able to be testedit can predict an outcomeit will explain the observationsall of these
Answers: 2

Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3

You know the right answer?
How many grams of solid iron will be produced from 32.1 grams of iron (III) oxide? Hint: Balance the...
Questions

Mathematics, 25.03.2021 14:00

English, 25.03.2021 14:00

Mathematics, 25.03.2021 14:00



Chemistry, 25.03.2021 14:00

Mathematics, 25.03.2021 14:00

English, 25.03.2021 14:00

Health, 25.03.2021 14:00


English, 25.03.2021 14:00


Biology, 25.03.2021 14:00

Chemistry, 25.03.2021 14:00

English, 25.03.2021 14:00

Law, 25.03.2021 14:00

Advanced Placement (AP), 25.03.2021 14:00

Mathematics, 25.03.2021 14:00

Mathematics, 25.03.2021 14:00

Mathematics, 25.03.2021 14:00