
Chemistry, 03.05.2021 18:50 datgamer13
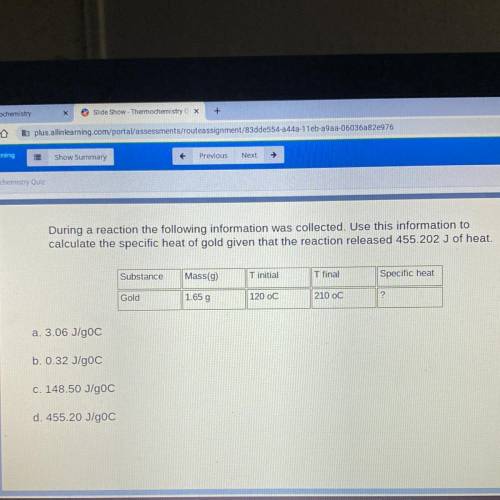
During a reaction the following information was collected. Use this information to
calculate the specific heat of gold given that the reaction released 455.202 J of heat.
Substance
Mass(9)
T initial
T final
Specific heat
Gold
1.65 g
120 oC
210 OC
2
a. 3.06 J/gOC
b. 0.32 J/goC
c. 148.50 J/goC
d. 455.20 J/goC

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 1

Chemistry, 22.06.2019 14:30
An object resting on a table weighs 100 n. with what force is the object pushing on the table? with what force is the table pushing on the object? explain how you got your answer.
Answers: 3


Chemistry, 22.06.2019 23:30
The ammonia molecule in the diagram has the observed bond orientation because
Answers: 1
You know the right answer?
During a reaction the following information was collected. Use this information to
calculate the s...
Questions

Mathematics, 22.04.2021 02:00

Mathematics, 22.04.2021 02:00


Mathematics, 22.04.2021 02:00




Physics, 22.04.2021 02:00

Mathematics, 22.04.2021 02:00

Mathematics, 22.04.2021 02:00



Mathematics, 22.04.2021 02:00


Mathematics, 22.04.2021 02:00

Business, 22.04.2021 02:00

English, 22.04.2021 02:00

Arts, 22.04.2021 02:00


Mathematics, 22.04.2021 02:00



