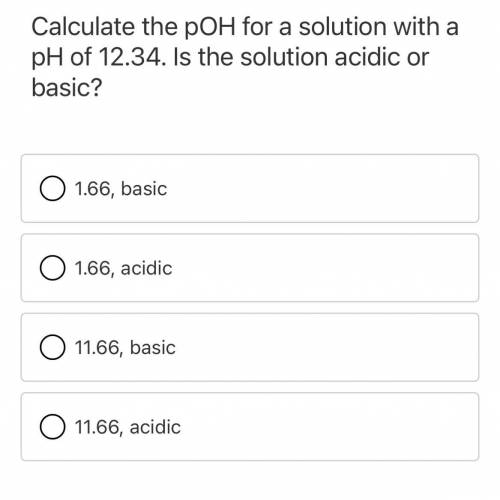
Calculate the pOH for a solutions with a pH of 12.34. Is the solution acidic or basic?
...

Chemistry, 04.05.2021 16:00 kendrabrown41
Calculate the pOH for a solutions with a pH of 12.34. Is the solution acidic or basic?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:50
Significant figures are digits read directly from the measuring instrument plus one more digit, which is __ by the observer.
Answers: 2

Chemistry, 22.06.2019 06:30
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2


Chemistry, 22.06.2019 16:50
What is conserved in the reaction shown below? h2(g) + cl2 (g) --> 2hcl(g)a. mass onlyb. mass and moles onlyc. mass, moles, and molecules onlyd. mass, moles, molecules, and volume
Answers: 2
You know the right answer?
Questions



Mathematics, 14.10.2019 01:00

Mathematics, 14.10.2019 01:00



Mathematics, 14.10.2019 01:00

English, 14.10.2019 01:00


Geography, 14.10.2019 01:00


Mathematics, 14.10.2019 01:00

History, 14.10.2019 01:00

History, 14.10.2019 01:00


Social Studies, 14.10.2019 01:00

Biology, 14.10.2019 01:00


Mathematics, 14.10.2019 01:00

Geography, 14.10.2019 01:00



