Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 07:30
11. phosphorus-32 is radioactive and has a half life of 14 days. how much of a 124 mg sample of phosphorus-32 is present after 56 days? a) 7.75 mg b) 15.5 mg c) 31.0 mg d) 62.0 mg
Answers: 3

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3

Chemistry, 22.06.2019 20:30
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3
You know the right answer?
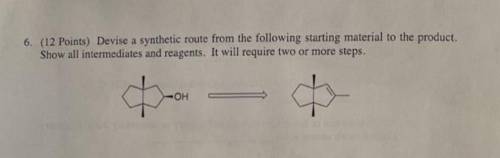
Devise a synthetic route from the following starting material to the product. Show all intermediates...
Questions

Mathematics, 17.02.2021 18:50

History, 17.02.2021 18:50

Advanced Placement (AP), 17.02.2021 18:50




History, 17.02.2021 18:50

Mathematics, 17.02.2021 18:50



English, 17.02.2021 18:50

Mathematics, 17.02.2021 18:50

Business, 17.02.2021 18:50


Chemistry, 17.02.2021 18:50



Mathematics, 17.02.2021 18:50

Mathematics, 17.02.2021 18:50