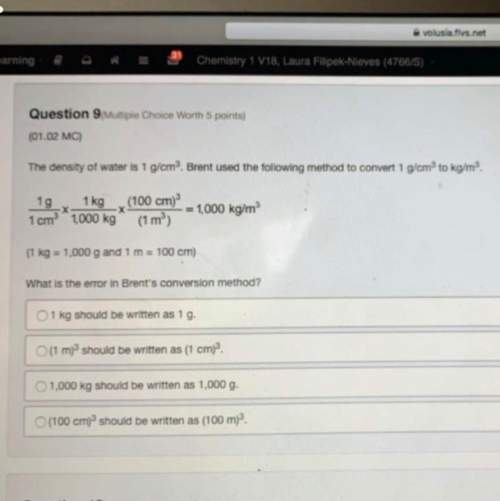
A 72.0-gram piece of metal at 96.0°C is placed in 130.0 g of water in a calorimeter at 25.5 °C. The
final temperature in the calorimeter is 31.0 °C. Determine the specific heat of the metal. Show your
work by listing various steps, and explain how the law of conservation of energy applies to this
situation. (8 points)

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:10
The concept of empiricism states that all rationally accepted knowledge is determined from experience. francis bacon was one of the first scientists to promote this theory. what was it’s impact on society?
Answers: 1

Chemistry, 21.06.2019 23:40
If the atomic mass of an atom is 34 and the atom contains 13 protons, how many neutrons does the atom contain?
Answers: 2

Chemistry, 22.06.2019 10:30
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1
You know the right answer?
A 72.0-gram piece of metal at 96.0°C is placed in 130.0 g of water in a calorimeter at 25.5 °C. The...
Questions

Health, 05.10.2019 08:00

History, 05.10.2019 08:00






Spanish, 05.10.2019 08:00



Business, 05.10.2019 08:00


Computers and Technology, 05.10.2019 08:00

Geography, 05.10.2019 08:00

Mathematics, 05.10.2019 08:00

Mathematics, 05.10.2019 08:00

Spanish, 05.10.2019 08:00

English, 05.10.2019 08:00

Mathematics, 05.10.2019 08:00

Advanced Placement (AP), 05.10.2019 08:00