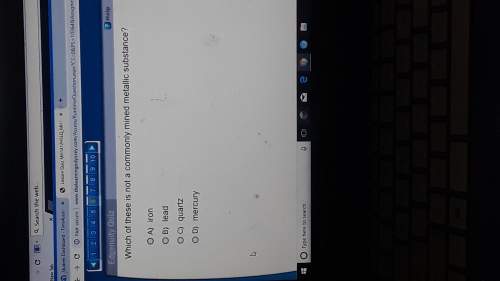
Chemistry, 07.05.2021 22:00 bartlettcs9817
Which statement best describes the relationship between an object's density and its index of refraction?
As the optical density increases, the refractive index increases.
As the mass per unit volume of the substance increases, the refractive index decreases.
As the mass of the object increases, the refractive index decreases.
As the physical density of the substance increases, the refractive index increases.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:30
Match the following vocabulary terms to their definitions. 1. amount of energy required to change 1 gram of material from the solid to the liquid state at its melting point 2. a measure of the kinetic energy of the particles of a substance 3. the amount of heat energy required to raise the temperature of 1 gram of liquid water from 14.5°c to 15.5°c 4. amount of energy required to change 1 gram of material from the liquid to the gaseous state at its boiling point 5. the amount of energy required to change 1 gram of a substance 1°c a. temperature b. latent heat of vaporization c. latent heat of fusion d. calorie e. specific heat
Answers: 1

Chemistry, 22.06.2019 06:40
Ted and emily played a mixed doubles tennis match against jack and brenda. in the second match. ted and brenda played against jack and emily. which type of chemical reaction does the situation demonstrate?
Answers: 3

Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 1

Chemistry, 22.06.2019 21:50
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1
You know the right answer?
Which statement best describes the relationship between an object's density and its index of refract...
Questions

English, 27.10.2020 08:50


Advanced Placement (AP), 27.10.2020 08:50

Chemistry, 27.10.2020 08:50


Mathematics, 27.10.2020 08:50

English, 27.10.2020 08:50

English, 27.10.2020 08:50

Physics, 27.10.2020 08:50


Mathematics, 27.10.2020 08:50

Mathematics, 27.10.2020 08:50


Biology, 27.10.2020 08:50

Biology, 27.10.2020 08:50

Arts, 27.10.2020 08:50

Mathematics, 27.10.2020 08:50

Social Studies, 27.10.2020 08:50