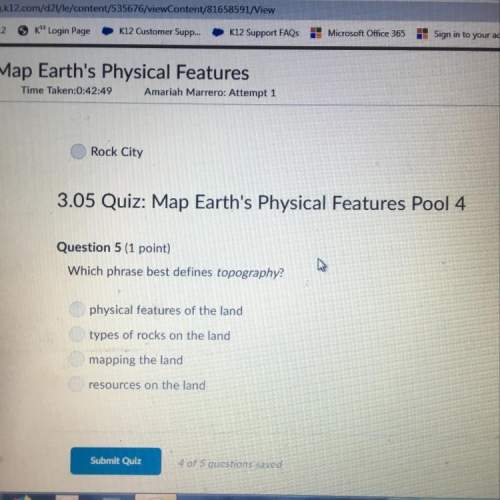
Desiré was investigating a chemical reaction. When she heated it up, she found that sulfuric acid changed into water. She made the following atomic-scale model to show what she thinks happened. Do you think this is a complete model of what happened during the chemical reaction?


Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:30
In saturated organic compounds, all the bonds between carbon atoms are called?
Answers: 1

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 17:10
)benzene and toluene form nearly ideal solutions. consider an equimolar solution of benzene and toluene. at 20 °c the vapour pressures of pure benzene and toluene are 9.9 kpa and 2.9 kpa, respectively. the solution is boiled by reducing the external pressure below the vapour pressure. calculate (i) the pressure when boiling begins, (ii) the composition of each component in the vapour, and (iii) the vapour pressure when only a few drops of liquid remain. assume that the rate of vaporization is low enough for the temperature to remain constant at 20 °c.
Answers: 1
You know the right answer?
Desiré was investigating a chemical reaction. When she heated it up, she found that sulfuric acid ch...
Questions




Biology, 16.10.2019 12:30

Chemistry, 16.10.2019 12:30




Mathematics, 16.10.2019 12:30

Chemistry, 16.10.2019 12:30

English, 16.10.2019 12:30

Mathematics, 16.10.2019 12:30




English, 16.10.2019 12:30



Chemistry, 16.10.2019 12:30

Chemistry, 16.10.2019 12:30