
Chemistry, 11.05.2021 18:50 1r32tgy5hk7
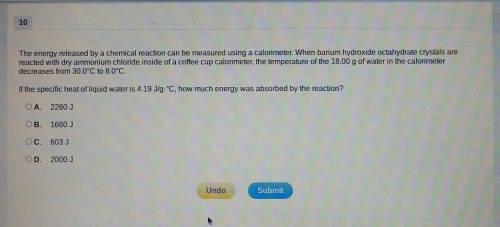
The energy released by a chemical reaction can be measured using a calorimeter. When barium hydroxide octahydrate crystals are reacted with dry ammonium chloride inside of a coffee cup calorimeter, the temperature of the 18.00g of water in the calorimeter decreases from 30.0⁰C to 8.0⁰C. If the specific heat of liquid water is 4.19J/g.⁰C, how much energy was absorbed by the reaction?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Which mathematical relationship should you us to convert moles of a substance into grams
Answers: 1

Chemistry, 21.06.2019 23:00
A100-watt light bulb radiates energy at a rate of 100 j/s. (the watt, a unit of power or energy over time, is defined as 1 j/s.) if all of the light emitted has a wavelength of 525 nm , how many photons are emitted per second?
Answers: 1

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

You know the right answer?
The energy released by a chemical reaction can be measured using a calorimeter. When barium hydroxid...
Questions


English, 10.07.2019 11:30



English, 10.07.2019 11:30



English, 10.07.2019 11:30

Mathematics, 10.07.2019 11:30


Mathematics, 10.07.2019 11:30







Physics, 10.07.2019 11:30




