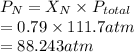Chemistry, 12.05.2021 04:30 irabrooks14
To lower the chance of suffering from decompression sickness (the bends), scuba divers use a mixture of gases in their air tank (typically oxygen and nitrogen gas in recreational dives). Assuming no other gas is present besides oxygen and nitrogen, if the mole fraction of oxygen present is 0.21, what is the partial pressure of nitrogen gas if the total pressure is 111.7 atm

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Llama have 74 chromosomes how many chromosomes will they be found in their gametes explain how you know
Answers: 2

Chemistry, 22.06.2019 20:10
What would happen to a volleyball left outside in the winter? o o o o a. it would expand. b. it would lose air. c. it would shrink. d. it would explode.
Answers: 2

Chemistry, 23.06.2019 00:30
How many moles of co2 are produced during the complete combustion of 3.6 moles of c2h6
Answers: 1

Chemistry, 23.06.2019 08:50
Reacting masses1 calcium carbonate breaks down on heating to produce calcium oxide and carbondioxide gas.caco3 + cao + co2a student heats 15 g of calcium carbonate strongly in a crucible.relative atomic masses (a): ca = 40, c = 12, o = 16.calculate the mass of calcium oxide produced by this reaction.(5 marks)
Answers: 3
You know the right answer?
To lower the chance of suffering from decompression sickness (the bends), scuba divers use a mixture...
Questions


Mathematics, 15.11.2019 19:31



Business, 15.11.2019 19:31

Medicine, 15.11.2019 19:31







Mathematics, 15.11.2019 19:31


English, 15.11.2019 19:31


Mathematics, 15.11.2019 19:31


Mathematics, 15.11.2019 19:31

History, 15.11.2019 19:31

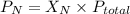
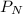 = partial pressure of nitrogen
= partial pressure of nitrogen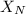 = mole fraction of nitrogen
= mole fraction of nitrogen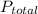 = total pressure
= total pressure