
Chemistry, 13.05.2021 03:30 hahalol123goaway
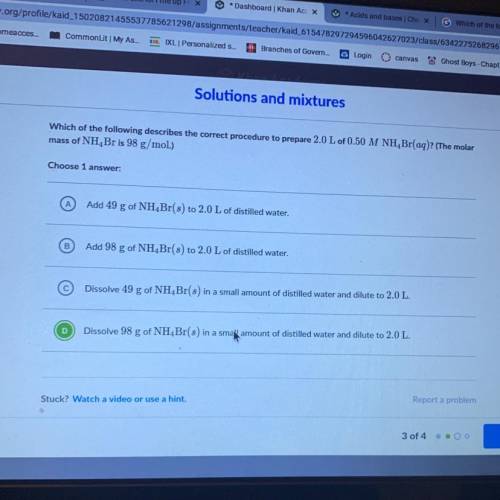
Which of the following describes the correct procedure to prepare 2.0 L of 0.50 M NH Br(aq)? (The molar
mass of NH, Br is 98 g/mol.)
Choose 1
BO
Add 49 g of NH Br(s) to 2.0 L of distilled water.
Add 98 g of NH Br(s) to 2.0 L of distilled water.
MY
Col
Dissolve 49 g of NH Br(s) in a small amount of distilled water and dilute to 2.0 L.
SA
Dissolve 98 g of NH Br(s) in a small amount of distilled water and dilute to 2.0 L.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:30
(1.6 × 10-19)(5.0 × 106) = c × 10d identify the missing numbers below to show the result of multiplying the numbers.
Answers: 1

Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium chlorate
Answers: 3

Chemistry, 23.06.2019 06:20
Examine the false statement. compounds are the smallest unit of an element that occur most commonly in nature. select the rewording of the statement that is true. a: atoms are the smallest unit of an element that commonly occur in nature. b: molecules are the smallest unit of an element or compound that commonly occur in nature. c: molecules are the smallest unit of a compound that occur on the periodic table. d: compounds are the smallest unit of an element that occur on the periodic table
Answers: 1

Chemistry, 23.06.2019 11:50
What is the oxidation half-reaction for this unbalanced redox equation? cr2o72– + fe2+ → cr3+ + fe3+ cr3+ → cr2o72– cr2o72– → cr3+ fe3+ → fe2+ fe2+ → fe3+?
Answers: 2
You know the right answer?
Which of the following describes the correct procedure to prepare 2.0 L of 0.50 M NH Br(aq)? (The mo...
Questions




Mathematics, 20.09.2019 19:30







Computers and Technology, 20.09.2019 19:30

English, 20.09.2019 19:30


History, 20.09.2019 19:30


Mathematics, 20.09.2019 19:30


History, 20.09.2019 19:30




