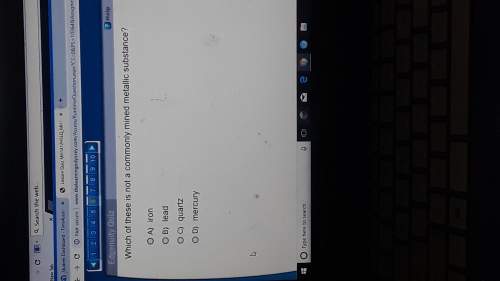
Chemistry, 15.05.2021 01:00 haydencheramie
How is the law of conservation of mass used to balance a chemical equation?
In a chemical equation, the products formed have the same atoms as the original reactants; however, the atoms may be rearranged. The law of conservation of mass states that the coefficients of atoms in the products must be equal to the coefficients of atoms in the reactants. Therefore, coefficients are used to balance the equation.
In a chemical equation, the products formed have the same atoms as the original reactants; however, the atoms may be rearranged. The law of conservation of mass states that the subscripts of atoms in the products must be equal to the subscripts of atoms in the reactants. Therefore, subscripts are used to balance the equation.
In a chemical equation, the products formed have the same atoms as the original reactants; however, the atoms may be rearranged. The law of conservation of mass states that the number of atoms of each element in the products must be equal to the number of atoms of the same element in the reactants. Therefore, subscripts are used to balance the equation.
In a chemical equation, the products formed have the same atoms as the original reactants; however, the atoms may be rearranged. The law of conservation of mass states that the number of atoms of each element in the products must be equal to the number of atoms of the same element in the reactants. Therefore, subscripts are used to balance the equation.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 11:30
Which statement best describes the flow of energy in this scenario
Answers: 1

Chemistry, 22.06.2019 19:10
Which statement correctly describes the phosphate ion, ? it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge on the phosphorus atom. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge on the phosphorus atom.
Answers: 3


Chemistry, 22.06.2019 21:30
How many oxygen atoms are there in 3.15 moles of hcl manganese (iv) oxide, mno2
Answers: 2
You know the right answer?
How is the law of conservation of mass used to balance a chemical equation?
In a chemical equation,...
Questions

English, 25.07.2019 13:00



History, 25.07.2019 13:00

Geography, 25.07.2019 13:00



Mathematics, 25.07.2019 13:00

Spanish, 25.07.2019 13:00



Mathematics, 25.07.2019 13:00

History, 25.07.2019 13:00

English, 25.07.2019 13:00


History, 25.07.2019 13:00

Mathematics, 25.07.2019 13:00

Mathematics, 25.07.2019 13:00

Mathematics, 25.07.2019 13:00