Chemistry, 15.05.2021 14:00 mathman783
Calculate the amount of 0.1 M base needed to neutralize 10,000 liters of pH 6.0 water. The base is Sodium Hydroxide (NaOH) Please show work!

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 15:20
Both 1,2−dihydronaphthalene and 1,4−dihydronaphthalene may be selectively hydrogenated to 1,2,3,4−tetrahydronaphthalene. one of these isomers has a heat of hydrogenation of 101 kj/mol (24.1 kcal/mol), and the heat of hydrogenation of the other is 113 kj/mol (27.1 kcal/mol). match the heat of hydrogenation with the appropriate dihydronaphthalene.
Answers: 2

Chemistry, 21.06.2019 22:20
Which of the following statements is false regarding aromaticity? a. the compound must be cyclic b. the compound must be fully conjugated c. the compound must be planar d.the number of electrons in the pi system must satisfy the hückel 4n+2 rule e. the compound must have a neutral charge
Answers: 2

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3
You know the right answer?
Calculate the amount of 0.1 M base needed to neutralize 10,000 liters of pH 6.0 water.
The base is...
Questions

Mathematics, 27.03.2020 20:35

Mathematics, 27.03.2020 20:35

Mathematics, 27.03.2020 20:35



Physics, 27.03.2020 20:35

Mathematics, 27.03.2020 20:36


Engineering, 27.03.2020 20:36


History, 27.03.2020 20:36

Chemistry, 27.03.2020 20:36


Mathematics, 27.03.2020 20:38



Mathematics, 27.03.2020 20:39


English, 27.03.2020 20:39

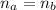
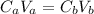
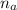 : is the number of moles of the acid
: is the number of moles of the acid 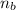 : is the number of moles of the base
: is the number of moles of the base 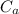 : is the concentration of the acid
: is the concentration of the acid 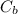 : is the concentration of the base = 0.1 M
: is the concentration of the base = 0.1 M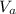 : is the volume of the acid = 10000 L
: is the volume of the acid = 10000 L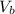 : is the volume of the base =?
: is the volume of the base =?![pH = -log([H^{+}])](/tpl/images/1326/0955/0d4b9.png)
![[H^{+}] = 10^{-pH} = 10^{-6} M](/tpl/images/1326/0955/c9161.png)