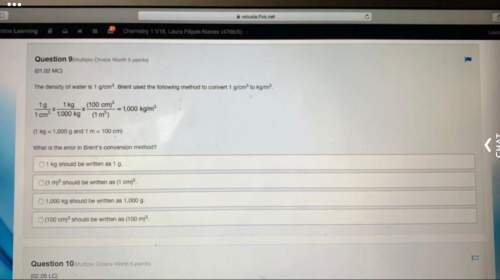
15.(06.04 LC)
What is the mass of 22.4 L of H2 at STP? (4 points)
01.01 gram
02.02 gram...

Chemistry, 17.05.2021 01:50 sheiladenson3619
15.(06.04 LC)
What is the mass of 22.4 L of H2 at STP? (4 points)
01.01 gram
02.02 grams
O 11.2 grams
m
22.4 grams

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:50
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1

Chemistry, 22.06.2019 03:10
Between 2014 and 2016, more than 25,000 children in flint, michigan, drank water that was contaminated with lead from lead pipes. during this time, the city claimed the water was safe to drink. which of these actions could the city have taken to ensure that the drinking water was free from lead?
Answers: 3

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1
You know the right answer?
Questions


Social Studies, 27.01.2020 22:31

English, 27.01.2020 22:31

Mathematics, 27.01.2020 22:31

Chemistry, 27.01.2020 22:31

English, 27.01.2020 22:31


Biology, 27.01.2020 22:31

Social Studies, 27.01.2020 22:31


Chemistry, 27.01.2020 22:31

History, 27.01.2020 22:31



Computers and Technology, 27.01.2020 22:31



Mathematics, 27.01.2020 22:31

Mathematics, 27.01.2020 22:31