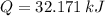Chemistry, 18.05.2021 14:00 fireemblam101ovu1gt
Calculate the total energy (in kJ) absorbed when 50.5 g of ice at -15.0°C is converted into liquid water at 65.0 °C.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 18:00
How is energy related to the change of state represented by the model? atoms gain energy as a solid changes to a liquid. atoms gain energy as a solid changes to a gas. atoms lose energy as a solid changes to a liquid. atoms lose energy as a solid changes to a gas.
Answers: 3


Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1
You know the right answer?
Calculate the total energy (in kJ) absorbed when 50.5 g of ice at -15.0°C is converted into liquid w...
Questions






Mathematics, 10.10.2019 17:10


Arts, 10.10.2019 17:10

English, 10.10.2019 17:10


Mathematics, 10.10.2019 17:10






Computers and Technology, 10.10.2019 17:10



![Q = m\cdot [c_{i}\cdot (T_{2}-T_{1})+L_{f} + c_{w}\cdot (T_{3}-T_{2})]](/tpl/images/1330/5123/31632.png) (1)
(1)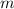 - Mass of the ice, in kilograms.
- Mass of the ice, in kilograms.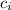 - Specific heat of ice, in kilojoules per kilogram-degree Celsius.
- Specific heat of ice, in kilojoules per kilogram-degree Celsius.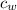 - Specific heat of water, in kilojoules per kilogram-degree Celsius.
- Specific heat of water, in kilojoules per kilogram-degree Celsius.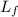 - Latent heat of fusion, in kilojoules per degree Celsius.
- Latent heat of fusion, in kilojoules per degree Celsius. 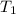 - Initial temperature of water, in degrees Celsius.
- Initial temperature of water, in degrees Celsius.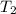 - Fusion point of water, in degrees Celsius.
- Fusion point of water, in degrees Celsius.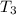 - Final temperature of water, in degree Celsius.
- Final temperature of water, in degree Celsius.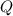 - Total energy absorbed, in kilojoules.
- Total energy absorbed, in kilojoules.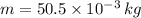 ,
, 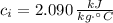 ,
, 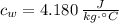 ,
, 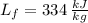 ,
, 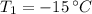 ,
, 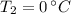 and
and 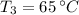 , then the total energy absorbed is:
, then the total energy absorbed is:![Q= (50.5\times 10^{-3}\,kg)\cdot \left[\left(2.090\,\frac{kJ}{kg\cdot ^{\circ}C} \right)\cdot (15\,^{\circ}C) + 334\,\frac{kJ}{kg}+ \left(4.180\,\frac{kJ}{kg\cdot ^{\circ}C} \right)\cdot (65\,^{\circ}C)\right]](/tpl/images/1330/5123/d15d0.png)