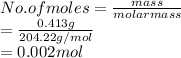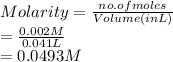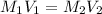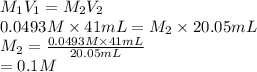Chemistry, 18.05.2021 19:20 christinamonte122
413 mg of dried KHP is dissolved in 41 mL of distilled water and titrated with potassium hydroxide (KOH). If it took 20.05 mL of KOH to reach the endpoint, determine the concentration of KOH. Show calculations.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 18:50
Which of the following is a conclusion that resulted from ernest rutherford’s scattering experiment? (will mark brainliest) a. the nucleus is negatively charged b. the atom is a dense solid and is indivisible c. the mass is conserved when atoms react chemically d. the nucleus is very small and the atom is mostly empty space
Answers: 3

Chemistry, 22.06.2019 19:30
What is the area in square meters of 448 g ai foil that has a thickness of 23921 nm? the density is 2.70 g/cm
Answers: 3

Chemistry, 22.06.2019 22:00
The volume of an unknown substance in a sealed glass jar is 50 milliliters. the volume of the jar is 200 milliliters. which state of matter could the substance be?
Answers: 2

You know the right answer?
413 mg of dried KHP is dissolved in 41 mL of distilled water and titrated with potassium hydroxide (...
Questions


Business, 09.03.2021 04:30

Physics, 09.03.2021 04:30








Mathematics, 09.03.2021 04:30



Mathematics, 09.03.2021 04:30

Mathematics, 09.03.2021 04:30

Mathematics, 09.03.2021 04:30


Physics, 09.03.2021 04:30


Mathematics, 09.03.2021 04:30