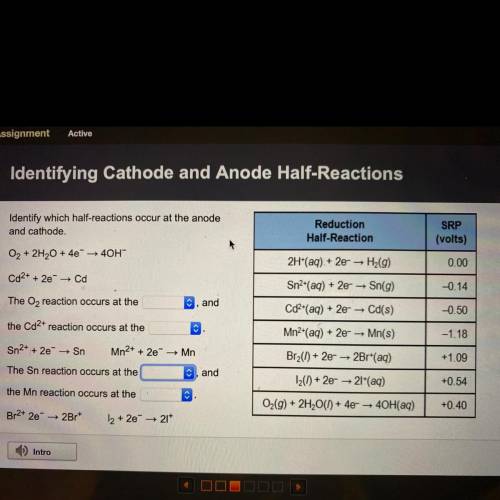
Identify which half-reactions occur at the anode
and cathode.
O2 + 2H2O + 4e + 40H-
Cd2...

Chemistry, 21.05.2021 01:00 ThatOneDumbAsian
Identify which half-reactions occur at the anode
and cathode.
O2 + 2H2O + 4e + 40H-
Cd2+ + 2e → Cd
The O2 reaction occurs at the
C, and
the Cd2+ reaction occurs at the
Sn2+ + 2e → Sn
Mn2+ + 2e → Mn
The Sn reaction occurs at the
, and

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:40
Astudent is given a sample of a blue copper sulfate hydrate. he weighs the sample in a dry covered porcelain crucible and got a mass of 23.875 g for the crucible, lid, and sample. the mass of the empty crucible and lid was found earlier to be 22.652 g. he then heats the crucible to expel the water of hydration, keeping the crucible at red heat for 10 minutes with the lid slightly ajar. on colling, he finds the mass of crucible, lid, and contents to be 23.403 g. the sample was changed in the process to very light clue anhydrous cuso4. if there are again 100.0 g of hydrate, how many grams of cuso4 are in it? how many moles of cuso4? (hint: molar mass of cuso4 = 159.6 g / mole. what per cent of the hydrate is cuso4? you may convert the mass of cuso4 to moles.)
Answers: 3

Chemistry, 22.06.2019 14:00
650.j is the same amount of energy as? 2720cal1550cal650.cal2.72cal
Answers: 2

Chemistry, 22.06.2019 21:30
What is the effect of returning nuclear reactor cooling water back into bodies of water?
Answers: 3

Chemistry, 22.06.2019 22:40
Covalent bonds generally form when the bonded elements have a difference in electronegativity less than 1.5. subtract the electronegativities for the following pairs of elements and predict whether they form a covalent bond. electronegativity difference of c and c: ionic covalent electronegativity difference of mg and cl: ionic covalent
Answers: 1
You know the right answer?
Questions


Biology, 19.05.2020 16:16


Mathematics, 19.05.2020 16:16


Mathematics, 19.05.2020 16:16

English, 19.05.2020 16:16

Mathematics, 19.05.2020 16:16


English, 19.05.2020 16:16



Mathematics, 19.05.2020 16:16

Mathematics, 19.05.2020 16:16

Mathematics, 19.05.2020 16:16


English, 19.05.2020 16:16

Chemistry, 19.05.2020 16:16




