
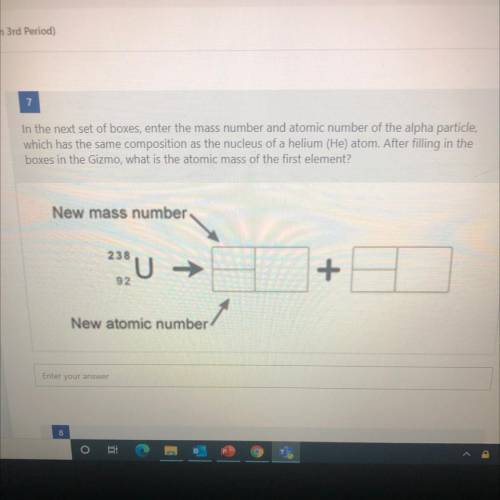
In the next set of boxes, enter the mass number and atomic number of the alpha particle,
which has the same composition as the nucleus of a helium (He) atom. After filling in the
boxes in the Gizmo, what is the atomic mass of the first element?
New mass number
238
U
+
92
New atomic number

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3

Chemistry, 22.06.2019 07:00
This image is an example of a(n) a) atom. b) compound. c) mixture. d) molecule.
Answers: 1

Chemistry, 22.06.2019 19:30
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2

Chemistry, 22.06.2019 21:20
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2
You know the right answer?
In the next set of boxes, enter the mass number and atomic number of the alpha particle,
which has...
Questions


Mathematics, 16.09.2019 05:30

English, 16.09.2019 05:30




Health, 16.09.2019 05:30

Biology, 16.09.2019 05:30


History, 16.09.2019 05:30

Mathematics, 16.09.2019 05:30

Health, 16.09.2019 05:30


Mathematics, 16.09.2019 05:30


Social Studies, 16.09.2019 05:30



History, 16.09.2019 05:30



