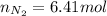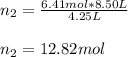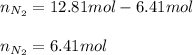Un recipiente cerrado, de 4,25 L, con tapa móvil, contiene H2S(g) a 740 Torr y 50,0°C. Se introduce en ese recipiente N2(g) a temperatura y presión constantes, de manera que el volumen final es el doble del volumen inicial. Calcular la cantidad de N2(g) en el recipiente, expresada en moles. porfi ayuda

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 3

Chemistry, 22.06.2019 18:50
Which of the following is a conclusion that resulted from ernest rutherford’s scattering experiment? (will mark brainliest) a. the nucleus is negatively charged b. the atom is a dense solid and is indivisible c. the mass is conserved when atoms react chemically d. the nucleus is very small and the atom is mostly empty space
Answers: 3

Chemistry, 22.06.2019 20:40
Select the correct value for the indicated bond angle in each of the compounds. o−o−oo−o−o angle of o3 90° 109.5° < 109.5° 120° < 120° 180° f−b−ff−b−f angle of bf3 180° < 109.5° < 120° 120° 109.5° 90° f−o−ff−o−f angle of of2 < 120° 120° 90° 109.5° 180° < 109.5° cl−be−clcl−be−cl angle of becl2 90° 109.5° 180° 120° < 109.5° < 120° f−p−ff−p−f angle of pf3 90° 109.5° < 109.5° 180° 120° < 120° h−c−hh−c−h angle of ch4 90° < 109.5° 180° 120° < 120° 109.5°
Answers: 1
You know the right answer?
Un recipiente cerrado, de 4,25 L, con tapa móvil, contiene H2S(g) a 740 Torr y 50,0°C. Se introduce...
Questions




Biology, 22.04.2020 13:09

Biology, 22.04.2020 13:10

Mathematics, 22.04.2020 13:10

Mathematics, 22.04.2020 13:12

Mathematics, 22.04.2020 13:12

English, 22.04.2020 13:13



Mathematics, 22.04.2020 13:18

Mathematics, 22.04.2020 13:19

Mathematics, 22.04.2020 13:19

History, 22.04.2020 13:19

Computers and Technology, 22.04.2020 13:19


Mathematics, 22.04.2020 13:19

History, 22.04.2020 13:19

Mathematics, 22.04.2020 13:19