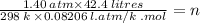Chemistry, 23.05.2021 14:00 ian2006huang
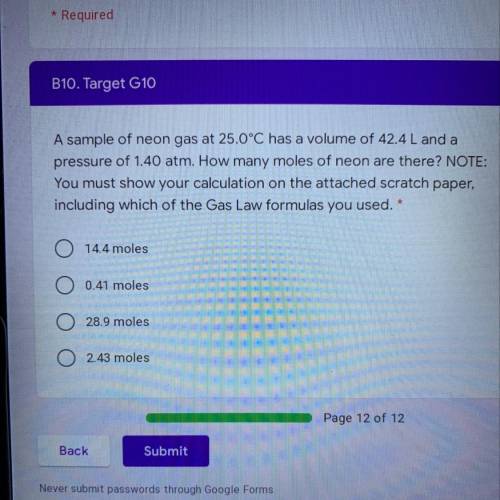
A sample of neon gas at 25.0°C has a volume of 42.4 L and a pressure of 1.40 atm. How many moles of neon are there? NOTE: You must show your calculation on the attached scratch paper,
including which of the Gas Law formulas you used. *
A. 14.4 moles
B. 0.41 moles
C. 28.9 moles
D. 2.43 moles
(Show how you did it please)

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 13:50
How does the motion of particles in a gas change as the gas cools
Answers: 2


Chemistry, 23.06.2019 02:40
Calculate the standard enthalpy of formation of liquid methanol, ch3oh(l), using the following information: c(graphite) + o2 latex: \longrightarrow ⟶ co2(g) latex: \delta δ h° = –393.5 kj/mol h2(g) + o2 latex: \longrightarrow ⟶ h2o(l) latex: \delta δ h° = –285.8 kj/mol ch3oh(l) + o2(g) latex: \longrightarrow ⟶ co2(g) + 2h2o(l) latex: \delta δ h° = –726.4 kj/mol
Answers: 3

Chemistry, 23.06.2019 03:30
Name atleast 3 type of energy associated with the microwave
Answers: 1
You know the right answer?
A sample of neon gas at 25.0°C has a volume of 42.4 L and a pressure of 1.40 atm. How many moles of...
Questions



Computers and Technology, 12.05.2021 21:40


SAT, 12.05.2021 21:40

Mathematics, 12.05.2021 21:40

Mathematics, 12.05.2021 21:40

Physics, 12.05.2021 21:40

Mathematics, 12.05.2021 21:40


Mathematics, 12.05.2021 21:40


Mathematics, 12.05.2021 21:40

Chemistry, 12.05.2021 21:40

English, 12.05.2021 21:40


Mathematics, 12.05.2021 21:40

Mathematics, 12.05.2021 21:40