Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3

Chemistry, 22.06.2019 22:30
Which of the following is not an assumption that scientists must make about the natural world? a. regularity b. causality c. predictability d. plausibility
Answers: 1

Chemistry, 23.06.2019 04:00
If you are told to get 100 ml of stock solution to use to prepare smaller size sample for an experiment, which piece of glassware would you use?
Answers: 3

Chemistry, 23.06.2019 08:40
The activation energy for this reaction is 75 kj·mol–1. the enzyme catalase (found in blood) lowers the activation energy to 8.0 kj·mol–1. at what temperature would the non-catalyzed reaction need to be run to have a rate equal to that of the enzyme-catalyzed reaction at 25°c?
Answers: 2
You know the right answer?
PLEASE PLEASE HELP
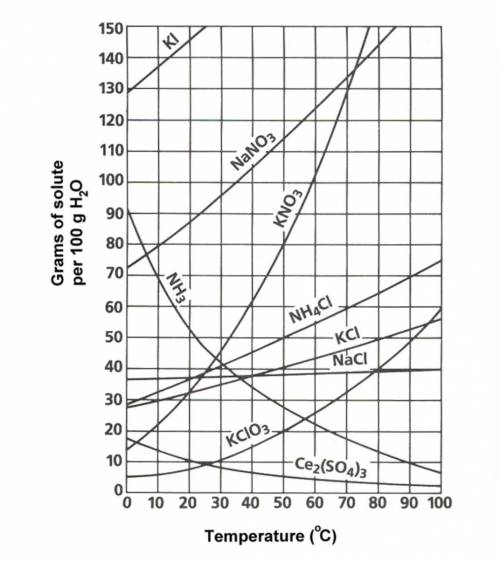
Approximately what mass of potassium nitrate will eventually precipitate from a...
Questions

Mathematics, 19.03.2021 06:50

Mathematics, 19.03.2021 06:50

Mathematics, 19.03.2021 06:50

Mathematics, 19.03.2021 06:50

History, 19.03.2021 06:50

English, 19.03.2021 06:50


Mathematics, 19.03.2021 06:50

Mathematics, 19.03.2021 06:50

Social Studies, 19.03.2021 06:50


English, 19.03.2021 06:50

Mathematics, 19.03.2021 06:50


English, 19.03.2021 06:50


Mathematics, 19.03.2021 06:50

Mathematics, 19.03.2021 06:50