
Chemistry, 25.05.2021 08:20 Christiancameron1234
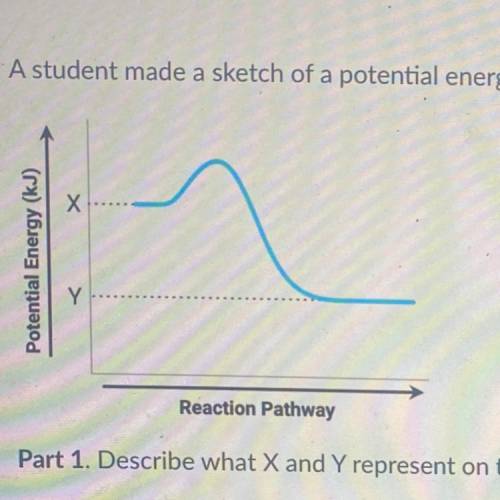
A student made a sketch of a potential energy diagram to represent an exothermic reaction.
Potential Energy (kJ)
Y
Reaction Pathway
Part 1. Describe what X and Y represent on the diagram.
Part 2. Explain how to determine the enthalpy change of a reaction on the diagram.
Part 3. Explain why the diagram made by the student is correct or incorrect.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:50
Problem page gaseous ethane reacts with gaseous oxygen gas to produce gaseous carbon dioxide and gaseous water . if of water is produced from the reaction of of ethane and of oxygen gas, calculate the percent yield of water. be sure your answer has the correct number of significant digits in it.
Answers: 2

Chemistry, 21.06.2019 22:00
If the particles in a sample of matter have an orderly arrangement and move only in place, the sample is a
Answers: 1


You know the right answer?
A student made a sketch of a potential energy diagram to represent an exothermic reaction.
Potentia...
Questions




Mathematics, 27.08.2019 02:00

Biology, 27.08.2019 02:00



Mathematics, 27.08.2019 02:00

Biology, 27.08.2019 02:00


Mathematics, 27.08.2019 02:00


Chemistry, 27.08.2019 02:00

Biology, 27.08.2019 02:00



Chemistry, 27.08.2019 02:00

History, 27.08.2019 02:00


Mathematics, 27.08.2019 02:00



