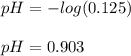Chemistry, 25.05.2021 21:20 martinbricein10
Given that ammonia (NH3) acts as a weak base in water with a Kb of 1.8 x 10-5 at 298.0 K, calculate the pH at 298.0 K of a solution prepared by mixing 100.0 mL of a 0.050 M aqueous solution of ammonia with 20.0 mL of a 1.00 M aqueous solution of nitric acid. The final volume of the solution is 120.0 mL.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:10
Building glycogen from glucose molecules is an example of
Answers: 3

Chemistry, 22.06.2019 12:20
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3

Chemistry, 22.06.2019 17:10
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1

Chemistry, 23.06.2019 08:40
The half-life of a certain element is 100 days. how many half-lives will it be before only one eighth of this elementremains?
Answers: 1
You know the right answer?
Given that ammonia (NH3) acts as a weak base in water with a Kb of 1.8 x 10-5 at 298.0 K, calculate...
Questions





Mathematics, 27.01.2021 14:00

Biology, 27.01.2021 14:00


Mathematics, 27.01.2021 14:00

Biology, 27.01.2021 14:00

Mathematics, 27.01.2021 14:00



History, 27.01.2021 14:00

Business, 27.01.2021 14:00




Social Studies, 27.01.2021 14:00

English, 27.01.2021 14:00

Computers and Technology, 27.01.2021 14:00

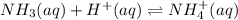
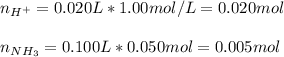
![[H^+]=\frac{0.015mol}{0.120L}=0.125M](/tpl/images/1347/3045/b1ea1.png)