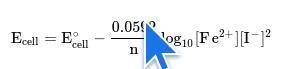Chemistry, 26.05.2021 20:40 jodiisbored2621
Use the given Nernst equation and reaction to solve this problem. What is the potential of this cell with the given conditions?
2Li (aq) + F2(g) 2Li+(aq) + 2F- (aq)
E° = +5.92 volts
T = 200°C
[Li+] = 10.0 molar
[F-] = 10.0 molar

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:50
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2

Chemistry, 22.06.2019 03:10
The covalent compound acetylene, which is the fuel of the oxyacetylene torch used by welders, has the molecular formula c2h2. the covalent compound benzene, a commercial solvent, has the molecular formula c6h6 each of these covalent compounds contains carbon and hydrogen atoms in a one-to-one ratio. would it be correct to write the chemical formulas of each as ch? explain.
Answers: 1

Chemistry, 22.06.2019 14:30
Connect the whole numbers on the periodic table to indicate what they represent?
Answers: 3

Chemistry, 23.06.2019 05:30
How many moles are in 1.26*10^24 particles in significant figures
Answers: 2
You know the right answer?
Use the given Nernst equation and reaction to solve this problem. What is the potential of this cell...
Questions


Mathematics, 06.01.2021 20:10

English, 06.01.2021 20:10

Mathematics, 06.01.2021 20:10

Mathematics, 06.01.2021 20:10


Health, 06.01.2021 20:10


English, 06.01.2021 20:10

Mathematics, 06.01.2021 20:10




Chemistry, 06.01.2021 20:10


Biology, 06.01.2021 20:10

Mathematics, 06.01.2021 20:10


Biology, 06.01.2021 20:10