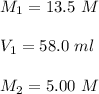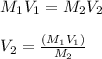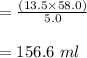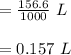Chemistry, 26.05.2021 20:50 dayanirisr45
A chemist must dilute of aqueous potassium iodide solution until the concentration falls to . He'll do this by adding distilled water to the solution until it reaches a certain final volume. Calculate this final volume, in liters. Round your answer to significant digits.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:00
Which atom or ion is the largest? a. k b. k+ c. ca d. ca2+ e. li
Answers: 1

Chemistry, 22.06.2019 10:30
Balance and in which category does it fit in? single or double displacement or synthesis or decomposition? (a) k2 o → k + o2 (b) na + i2 → nai (c) cu(no3 )2 + naoh → cu(oh)2 + nano3 (d) kclo3 → kcl + o2 (e) ca(no3 )2 + hbr → cabr2 + hno3 (f) sn(oh)2 → sno + h2 o (g) p4 + n2 o → p4 o6 + n2 (h) fe + al2 (so4 )3 → feso4 + al (i) alcl3 + na2 co3 → al2 (co3 )3 + nacl (j) c3 h6 + o2 → co2 + h2 o
Answers: 1


Chemistry, 22.06.2019 14:30
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2
You know the right answer?
A chemist must dilute of aqueous potassium iodide solution until the concentration falls to . He'll...
Questions

Biology, 06.05.2020 20:40

Biology, 06.05.2020 20:40


Biology, 06.05.2020 20:40

History, 06.05.2020 20:40

Mathematics, 06.05.2020 20:40







Medicine, 06.05.2020 20:40


English, 06.05.2020 20:40

Computers and Technology, 06.05.2020 20:41

Biology, 06.05.2020 20:41

Mathematics, 06.05.2020 20:41

Geography, 06.05.2020 20:41

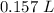 "
"