
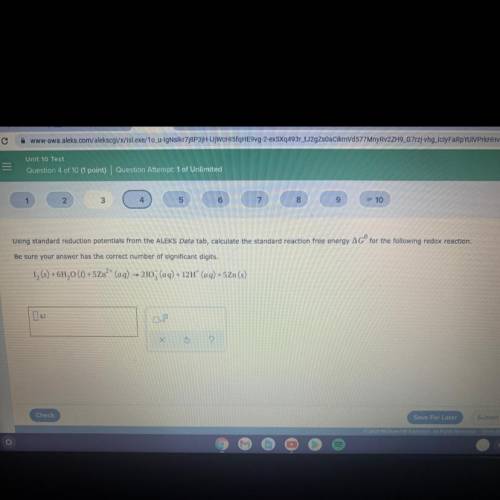
Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free energy AGº for the following redox reaction.
Be sure your answer has the correct number of significant digits.
12 (5) + 6H20 (1) + 5Zn²+ (aq) — 2103 (aq) + 12H+ (aq) +5Zn(s)

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:00
Nonpoint source pollution is difficult to control because it
Answers: 2

Chemistry, 22.06.2019 13:00
Using the thermodynamic information in the aleks data tab, calculate the standard reaction free energy of the following chemical reaction: →+p4o10s6h2ol4h3po4s round your answer to zero decimal places.
Answers: 3

Chemistry, 23.06.2019 06:00
In an exothermic reaction at equilibrium, what is the effect of lowering the temperature? a. the reaction makes more products. b. the reaction makes more reactants. c. the reaction is unchanged.
Answers: 1

Chemistry, 23.06.2019 11:30
If 4.8 moles of x and 3.4 moles of y react according to the reaction below, how many moles of the excess reactant will be left over at the end of the reaction? 3x + 2y “yields”/ x3y2. a. 1.7 mol y left over b. 1.6 mol x left over c. 0.2 mol y left over d. 0.1 mol x left over
Answers: 1
You know the right answer?
Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free en...
Questions



Mathematics, 10.12.2019 02:31

History, 10.12.2019 02:31


Mathematics, 10.12.2019 02:31


English, 10.12.2019 02:31

Mathematics, 10.12.2019 02:31


Mathematics, 10.12.2019 02:31

Mathematics, 10.12.2019 02:31

Mathematics, 10.12.2019 02:31

History, 10.12.2019 02:31

Social Studies, 10.12.2019 02:31

Mathematics, 10.12.2019 02:31

Mathematics, 10.12.2019 02:31


Spanish, 10.12.2019 02:31

History, 10.12.2019 02:31



