Consider the balanced reaction
below:
2Na + 2H2O → 2NaOH + H2
How many moles of sodium<...

Chemistry, 29.05.2021 07:00 Ciarrathereal
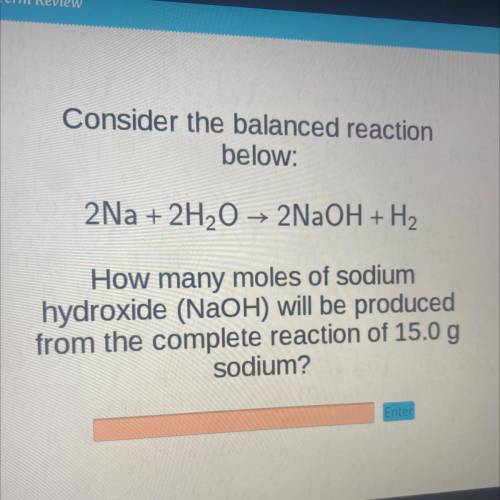
Consider the balanced reaction
below:
2Na + 2H2O → 2NaOH + H2
How many moles of sodium
hydroxide (NaOH) will be produced
from the complete reaction of 15.0 g
sodium?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:20
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 22.06.2019 00:00
Which of the following statements is true? a. elements in the last period are radioactive. b. atomic weight is the same as atomic mass. c. elements in the same group have the same number of electron shells. d. atomic number equals the number of neutrons in the nucleus of an atom.
Answers: 1

Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 04:30
How do i complete this electrolysis of water lab? i’m at home, so i don’t have the materials, and the lab didn’t properly work and was incomplete at school.
Answers: 1
You know the right answer?
Questions


Mathematics, 16.02.2020 21:16




Social Studies, 16.02.2020 21:17


English, 16.02.2020 21:19


Mathematics, 16.02.2020 21:21


Biology, 16.02.2020 21:22

Social Studies, 16.02.2020 21:22

Geography, 16.02.2020 21:22



Mathematics, 16.02.2020 21:26



Arts, 16.02.2020 21:26



