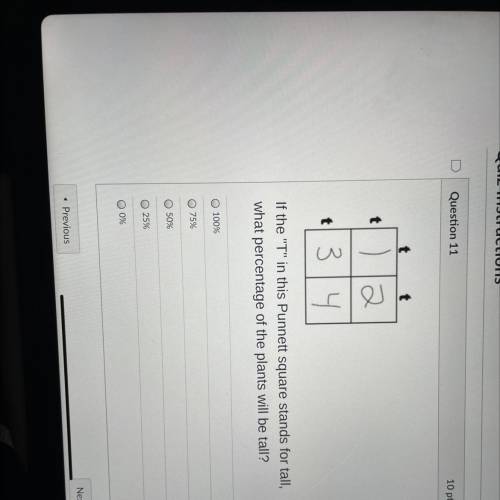
HELP………………….
...

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:20
Asolution is made by dissolving 25.5 grams of glucose (c6h12o6) in 398 grams of water. what is the freezing point depression of the solvent if the freezing point constant is -1.86 °c/m? show all of the work needed to solve this problem.
Answers: 1


Chemistry, 22.06.2019 02:40
The difference between the atomic number of an element and the element’s atomic mass is the number of ions.
Answers: 3

Chemistry, 22.06.2019 23:30
The density of the solid phase of a substance is 0.90 g/cm3 and the density of the liquid phase is 1.0 g/cm3. a large increase in pressure will a. lower the freezing point b. raise the freezing point c. lower the boiling point d. raise the triple point e. lower the triple point
Answers: 1
You know the right answer?
Questions

English, 31.01.2020 17:50

Mathematics, 31.01.2020 17:50



Advanced Placement (AP), 31.01.2020 17:50



Chemistry, 31.01.2020 17:50

Mathematics, 31.01.2020 17:50

Mathematics, 31.01.2020 17:50








History, 31.01.2020 17:50


Geography, 31.01.2020 17:50