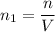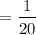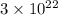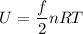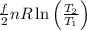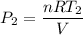A quarter of a mole of neon gas is in equilibrium at 330 K in a container that has a volume of 5.0L.
(a) How many atoms of neon would you expect to find in one portion of the container that has a volume of 1.0L? Explain your answer in terms of the definition of equilibrium given in our study of entropy.
(b) What is the thermal energy of the gas in this state?
(c) If the container is heated so that the gas reaches a temperature of 380 K, what is the change in the entropy of the system? Assume that the expansion of the container is negligible. (If you can give a number, do so; otherwise, at least say whether the entropy increases or decreases.)
(d) If the expansion of the container is negligible, what must the pressure of the heated gas be?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 14:20
Which of the following statements is not true? • a. covalent compounds have low melting and boiling points. • ob. covalent bonds between atoms of a compound are relatively weak compared to bonds between molecules. • c. covalent bonds occur between nonmetals. • d. covalent compounds are often gases or liquids.
Answers: 2

Chemistry, 21.06.2019 15:30
Which substance absorbs 58.16 kj of energy when 3.11 mol vaporizes? a)ch4 b)h2s c)co2 d)nacl
Answers: 2


Chemistry, 22.06.2019 01:00
Water is important for the of cells. a: size, shape, and temperature b: temperature, color, and odor c: color, odor, and size d: shape, temperature, and color
Answers: 2
You know the right answer?
A quarter of a mole of neon gas is in equilibrium at 330 K in a container that has a volume of 5.0L....
Questions

Mathematics, 27.10.2020 23:40

History, 27.10.2020 23:40

Mathematics, 27.10.2020 23:40

Mathematics, 27.10.2020 23:40

Mathematics, 27.10.2020 23:40

Health, 27.10.2020 23:40



Biology, 27.10.2020 23:40

Mathematics, 27.10.2020 23:40


Mathematics, 27.10.2020 23:40

Biology, 27.10.2020 23:40



Biology, 27.10.2020 23:40

Mathematics, 27.10.2020 23:40

Geography, 27.10.2020 23:40


Mathematics, 27.10.2020 23:40