
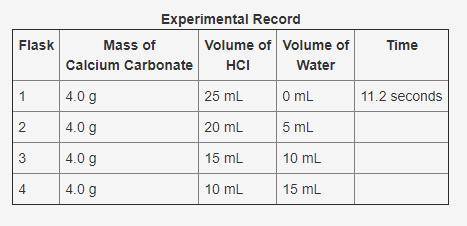
In an experiment, calcium carbonate reacted with different volumes of hydrochloric acid in water. One of the products formed during the experiment was carbon dioxide. The time taken for 0.89 mL of carbon dioxide to form was recorded. A partial record of the experiment is shown. Based on your knowledge of factors that affect the rates of chemical reactions, predict the trend in the last column of the experimental record. Use complete sentences to explain the trend you predicted. You do not have to determine exact values for time; just describe the trend you would expect (increase or decrease) and why it occurs.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:30
This drawing shows a human body system. what is the primary function of this body system?
Answers: 3

Chemistry, 22.06.2019 09:00
Chen drew a diagram to compare the ways in which different organisms obtain nitrogen. which label belongs to the area marked z?
Answers: 3

Chemistry, 22.06.2019 09:40
Apiece of copper has a temperature of 75.6 0c. when the metal is placed in 100.0 grams of water at 19.1 0c, the temperature rises by 5.5 0c. what is the mass of the metal?
Answers: 1

Chemistry, 22.06.2019 10:00
Miner's coal distributors does not mine coal itself, nor does it even store or handle the coal. instead, miner's solicits orders for low sulfur coal from other firms, then purchases the required amount from suppliers and directs them to ship the coal to its customers. what is miner's
Answers: 1
You know the right answer?
In an experiment, calcium carbonate reacted with different volumes of hydrochloric acid in water. On...
Questions

Mathematics, 14.10.2020 14:01






Mathematics, 14.10.2020 14:01

Mathematics, 14.10.2020 14:01

Business, 14.10.2020 14:01

Chemistry, 14.10.2020 14:01


Mathematics, 14.10.2020 14:01



Mathematics, 14.10.2020 14:01

Chemistry, 14.10.2020 14:01




Mathematics, 14.10.2020 14:01



