Chemistry, 01.06.2021 16:00 dededese2403
112 g of aluminum carbide react with 174 g water to produce methane and aluminum hydroxide in the reaction shown below.
2 Al4C3(s) + 12 H2O(l) → 3 CH4(g) + 4 Al(OH)3(s)
If aluminum carbide is the limiting reactant, how many moles of the excess reactant are left over
a
37.3 mol
b
4.68 mol
c
7.33 mol
d
131.94 mol

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:30
How air particles exert a pressure on the inside of the balloon
Answers: 1

Chemistry, 22.06.2019 10:00
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2


Chemistry, 22.06.2019 23:30
Aweight lifter raises a 1600 n barbell to a height of 2.0 meters. how much work was done? w = fd a) 30 joules b) 3000 joules c) 320 joules d) 3200 joules
Answers: 2
You know the right answer?
112 g of aluminum carbide react with 174 g water to produce methane and aluminum hydroxide in the re...
Questions


Mathematics, 23.10.2021 18:50




Mathematics, 23.10.2021 19:00



Mathematics, 23.10.2021 19:00

Mathematics, 23.10.2021 19:00

Physics, 23.10.2021 19:00

English, 23.10.2021 19:00



Social Studies, 23.10.2021 19:00





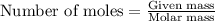 .....(1)
.....(1)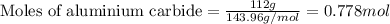
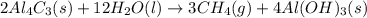
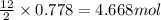 of water
of water