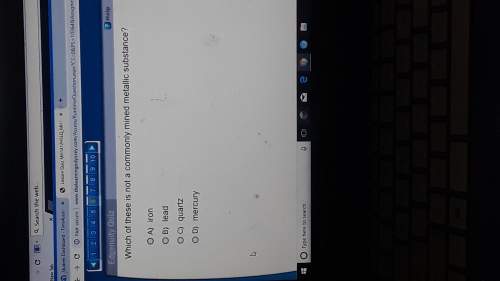
Chemistry, 02.06.2021 21:50 ramenbowlz
3. According to the solubility prediction for vitamin K1, the polar carbonyl functional groups significantly impact the solubility, presumably due to the orientation of the carbonyl bonds.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 01:10
Which of the following elements would you expect to have the lowest ionization energy value? fluorine, lithium, neon, nitrogen
Answers: 2

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2

Chemistry, 23.06.2019 04:20
The reaction below shows a system in equilibrium. how would a decrease in temperature affect this reaction? a. the rate of formation of the gases would increase. b. the equilibrium of the reaction would shift to the left. c. the equilibrium would shift to cause the gases to sublime into solids. d. the chemicals on the left would quickly form the chemical on the right.
Answers: 1
You know the right answer?
3. According to the solubility prediction for vitamin K1, the polar carbonyl functional groups sign...
Questions

Biology, 06.10.2019 06:20

History, 06.10.2019 06:20

Mathematics, 06.10.2019 06:20

Arts, 06.10.2019 06:20




Mathematics, 06.10.2019 06:20


Mathematics, 06.10.2019 06:20



Mathematics, 06.10.2019 06:20



English, 06.10.2019 06:20

Chemistry, 06.10.2019 06:20

Mathematics, 06.10.2019 06:20